RaDR Mice: Fluorescence Detection of Mutant Cells
 Homologous recombination is critical for repairing double strand breaks (DSBs), but it likely evolved primarily as amechanism for repairing broken replication forks. DNA replication forks can break down when they encounter DNA lesions.
Homologous recombination is critical for repairing double strand breaks (DSBs), but it likely evolved primarily as amechanism for repairing broken replication forks. DNA replication forks can break down when they encounter DNA lesions.
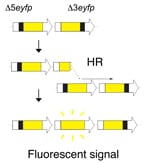 In order to detect homologous recombination in adult animals, we genetically engineered a DNA substrate wherein recombination between two non-functional copies of enhanced yellow fluorescent protein (EYFP) gives rise to fluorescent cells.
In order to detect homologous recombination in adult animals, we genetically engineered a DNA substrate wherein recombination between two non-functional copies of enhanced yellow fluorescent protein (EYFP) gives rise to fluorescent cells.
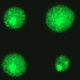
Rare recombinant cells can be seen in the pancreatic tissue of the fluorescent yellow direct repeat (FYDR).
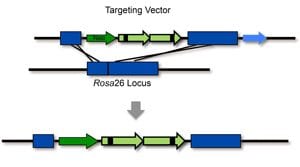
A limitation of FYDR mice is that many tissues do not express the construct, so fluorescence cannot be detected. We therefore targeted a recombination reporter to the Rosa26 locus, which is known to be highly expressed.
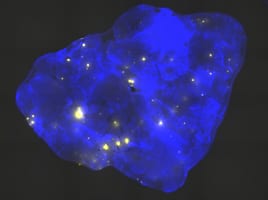
 Recombinant cells can be detected in almost all tissues of the Rosa26 direct repeat (RADR) mice.
Recombinant cells can be detected in almost all tissues of the Rosa26 direct repeat (RADR) mice.
The FYDR mice have been used extensively. Most recently, we have used these mice to probe the relationship between cell division and recombination in vivo, and to explore the impact of inflammation on recombination susceptibility.
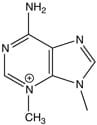
We are exploring the relationships between specific DNA lesions (such as 3-methyladenine, shown here) and homologous recombination events.